11.1. Coevolutionary interactions between insects and plants
Reciprocal interactions over evolutionary time be- tween phytophagous insects and their food plants, or between pollinating insects and the plants they pollinate, have been described as coevolution. This term, coined by P.R. Ehrlich and P.H. Raven in 1964 from a study of butterflies and their host plants, was defined broadly, and now several modes of coevolution are recognized. These differ in the emphasis placed on the specificity and reciprocity of the interactions.
Specific or pair-wise coevolution refers to the evolution of a trait of one species (such as an insect’s ability to detoxify a poison) in response to a trait of another species (such as the elaboration of the poison by the plant), which in turn evolved originally in response to the trait of the first species (i.e. the insect’s food preference for that plant). This is a strict mode of coevolution, as reciprocal interactions between specific pairs of species are postulated. The outcomes of such coevolution may be evolutionary “arms races” between eater and eaten, or convergence of traits in mutualisms so that both members of an interacting pair appear perfectly adapted to each other. Reciprocal evolution between the interacting species may contribute to at least one of the species becoming subdivided into two or more reproductively isolated populations (as exemplified by figs and fig wasps; Box 11.4), thereby generating species diversity.
Another mode, diffuse or guild coevolution, describes reciprocal evolutionary change among groups, rather than pairs, of species. Here the criterion of specificity is relaxed so that a particular trait in one or more species (e.g. of flowering plants) may evolve in response to a trait or suite of traits in several other species (e.g. as in several different, perhaps distantly related, pollinating insects).
These are the main modes of coevolution that relate to insect—plant interactions, but clearly they are not mutually exclusive. The study of such interactions is beset by the difficulty in demonstrating unequivocally that any kind of coevolution has occurred. Evolution takes place over geological time and hence the selection pressures responsible for changes in “coevolving” taxa can be inferred only retrospectively, principally from correlated traits of interacting organisms. Specificity of interactions among living taxa can be demonstrated or refuted far more convincingly than can historical reciprocity in the evolution of the traits of these same taxa. For example, by careful observation, a flower bearing its nectar at the bottom of a very deep tube may be shown to be pollinated exclusively by a particular fly or moth species with a proboscis of appropriate length (e.g. Fig. 11.8), or a hummingbird with a particular length and curvature of its beak. Specificity of such an association between any individual pollinator species and plant is an observable fact, but flower tube depth and mouthpart morphology are mere correlation and only suggest coevolution (section 11.3.1).
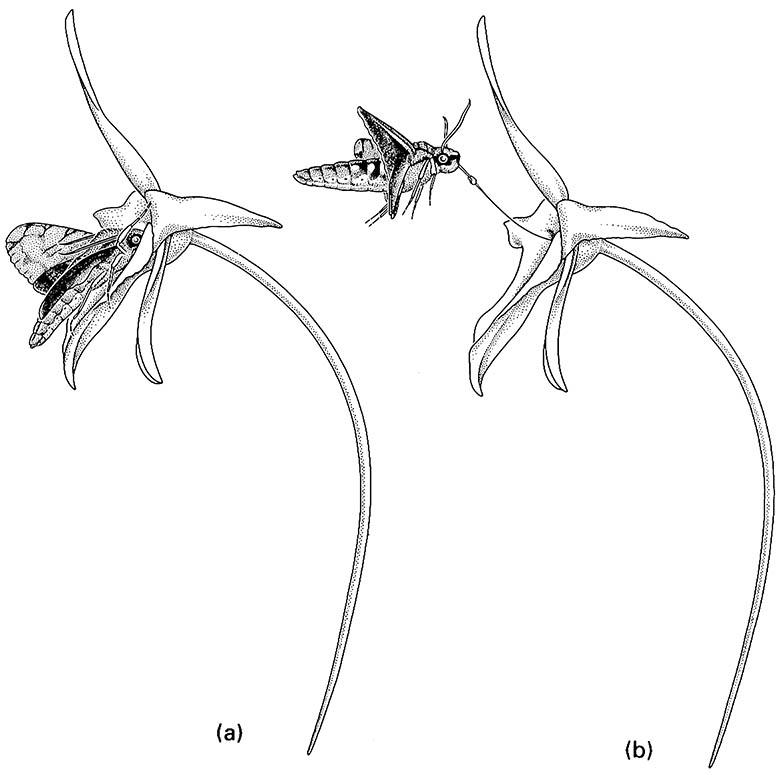
(a) full insertion of the moth’s proboscis; (b) upward flight during withdrawal of the proboscis with the orchid pollinium attached. (After Wasserthal 1997)

