8.4. Evolution of wings
As we have seen, much of the success of insects can be attributed to the wings, found in the numerically dominant pterygotes. Pterygotes are unusual among winged animals in that no limbs lost their pre-existing function as a result of the acquisition of flight. As we cannot observe the origins of flight, and fossils (although relatively abundant) have not greatly assisted in interpretation, any hypotheses of the origins of flight must be speculative. Several ideas have been promoted, and the area remains controversial.
One of the longest-standing hypotheses attributes the origin of the wings to postulated lobes, derivations from the thoracic terga, called paranota. These lobes were not articulated and thus tracheation, innervation, venation, and musculature would have been of secondary derivation. The paranotal lobe hypothesis has been displaced in favor of one inferring wing origination from serially repeated, pre-existing, mobile structures of the pleuron. These were most probably the outer appendage (exite) and inner appendage (endite) of a basal leg segment, the epicoxa (Fig. 8.4a). Each “protowing” or winglet was formed by the fusion of an exite and endite lobe of the respective ancestral leg, with exites and endites having tracheation and articulation. Fossil evidence indicates the presence of articulated winglets on all body segments, best developed on the thorax (Fig. 8.4b). Molecular studies of development (Box 6.1) seem to substantiate the exite—endite model for wing origins.
The exite—endite hypothesis of wing origin can be reconciled with another recurring view: that wings derive from tracheal gills of an ancestral aquatic “pro- topterygote”. Although the earliest insects undoubtedly were terrestrial, the earliest pterygote insects probably had aquatic immature stages (section 8.3). The abdominal gills of aquatic mayfly nymphs may be homologous with the abdominal winglets of the protopterygote, and be serially homologous with thoracic wings. Winglets are postulated in aquatic juveniles to have functioned in gas exchange and/or ventilation or even to assist in swimming, with the terrestrial adult co-opting them for an aerodynamic function.
All hypotheses concerning early wings make a common assumption that winglets originally had a non-flight function, as small winglets could have little or no use in flapping flight. Suggestions for preadaptive functions have included any (or all!) of the following:
- protection of the legs;
- covers for the spiracles;
- thermoregulation;
- sexual display;
- aid in concealment by breaking up the outline;
- predator avoidance by extension of escape jump by gliding.
Aerodynamic function came only after enlargement. However, aquatic nymphal gills may even have been large enough to give some immediate significant aerial advantage if retained in a terrestrial adult.
The manner in which flight evolved is also highly speculative and contentious but, whatever the origin of winglets, they came to assume some aerodynamic function. Four routes to flight have been argued, via:
- floating, in which small insects were assisted in passive dispersal by convection;
- paragliding, in which winglets assisted in stable gliding or parachuting from trees and tall vegetation;
- running—jumping to flying;
- surface sailing, in which the raised winglets allowed the adults of aquatic insects to skim across the water surface.
The first two hypotheses apply equally to fixed, non-articulated winglets and to articulated but rigidly extended winglets. Articulated winglets and flapping flight can most easily be incorporated into the running—jumping scenario of developing flight. The “floating” route to flight suffers from the flaw that wings actually hinder passive dispersal, and selection would tend to favor diminution in body size and reduction in the wings with commensurate increase in features such as long hairs. The third, running-jump route is unlikely, as no insect could attain the necessary velocity for flight originating from the ground, and only the scenario of a powered leap to allow limited gliding or flight is at all plausible.
The surface-sailing hypothesis requires articulated winglets and can account for the loss of the abdominal winglets, which would be downwind of the thoracic ones and thus barely contribute to sailing performance. Some extant stoneflies (Plecoptera) can sail across water in this manner. Perhaps surface sailing drove the evolution of wing length more than aerial gliding did, and when winglets had reached certain dimensions then gliding or flapping flight may have been facilitated greatly.
Aerodynamic theory has been applied to the problem of how large winglets had to be to give some aerodynamic advantage, and model insects have been constructed for wind-tunnel testing. Although a size-constrained and fixed-wing model lacks realism, the following evidence has been produced. An unwinged 1 cm-long insect model lacks control in a glide, and even small winglets give an immediate advantage by allowing some retarding of velocity. The possession of caudal filaments and/or paired cerci would give greater glide stability, particularly when associated with the reduction and eventual loss of posterior abdominal winglets. Additional control over gliding or flight would come with increase in body and winglet size.
There is a basic structural division of the ptergygotes into “Palaeoptera”, with movable, non-folding wings, and the Neoptera, with complex wing articulation that allows folding of the wings backwards along the body (section 7.4.2). Some authors have suggested that the two wing-base types are so different that wings must have originated at least twice. However, it can be demonstrated that there is a basic venational pattern common to all pterygotes irrespective of the articulation, implying monophyly (a single origin) of wings, but not necessarily of flight. The primitive pterygote wing base apparently involved many articulated sclerites: such a system is seen in fossil palaeopterans and, in a variably modified form, in extant neopterans. In the Ephemeroptera and Odonata the basal sclerites have undergone extensive fusion that prevents the wing from flexing backwards. However, the nature of these fusions, and others that have occurred within the neopteran lineages, indicates that many different path- ways have been used, and fusion per se does not indicate monophyly of the Palaeoptera. The likelihood that the primitive winglet had complex articulation provides a major criticism of the aerodynamic hypothesis above and the supposition of fixed wings renders suspect the conclusions from these unrealistic model insects.
The traditional proposal for the origin of venation involves tracheated, supporting or strengthening ridges on the protowing. Alternatively, the veins arose along the courses of hemolymph canals that supplied the winglets, in a manner seen in the gills of some aquatic insects. The basic venational pattern (section 2.4.2, Fig. 2.21) consists of eight veins each arising from a basal blood sinus, named from anterior to posterior: precosta, costa, subcosta, radius, media, cubitus, anal, and jugal. Each vein (perhaps excepting the media) branched basally into anterior concave and posterior convex components, with additional dichotomous branching away from the base, and a polygonal pattern of cells. Evolution of the insect wing has involved frequent reduction in the number of cells, development of bracing struts (cross-veins), selected increase in division of some veins, and reduction in complexity or complete loss of others. Furthermore, there have been changes in the muscles used for powered flight and in the phases of wing beat (section 3.1.4). Alteration in function has taken place, including the protection of the hind pair of wings by the modified fore wings (tegmina or elytra) in some groups. Increased flight control has been gained in some other groups by coupling the fore and hind wings as a single unit, and in Diptera by reduction of the metathoracic wings to halteres that function like gyroscopes.
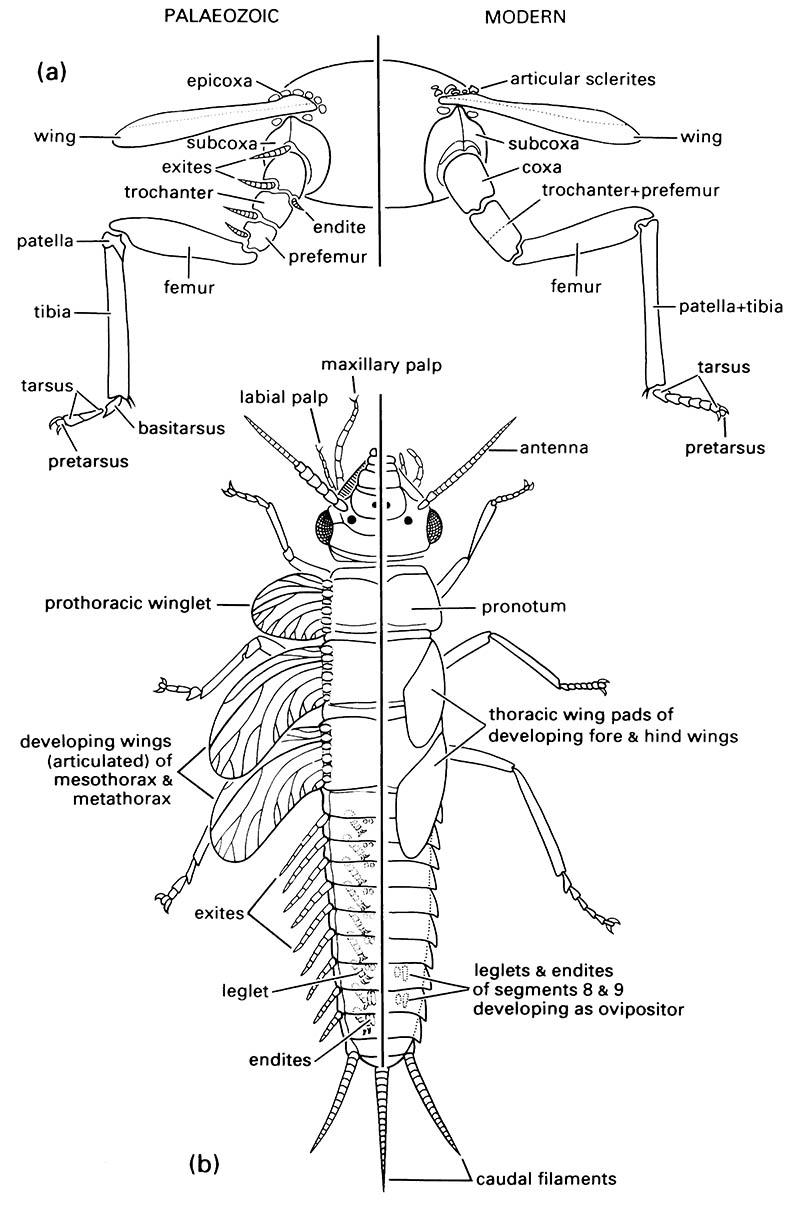
(a) thoracic segment of adult showing generalized condition of appendages; (b) dorsal view of nymphal morphology. (Modified from Kukalová-Peck 1991; to incorporate ideas of J.W.H. Trueman (unpublished))
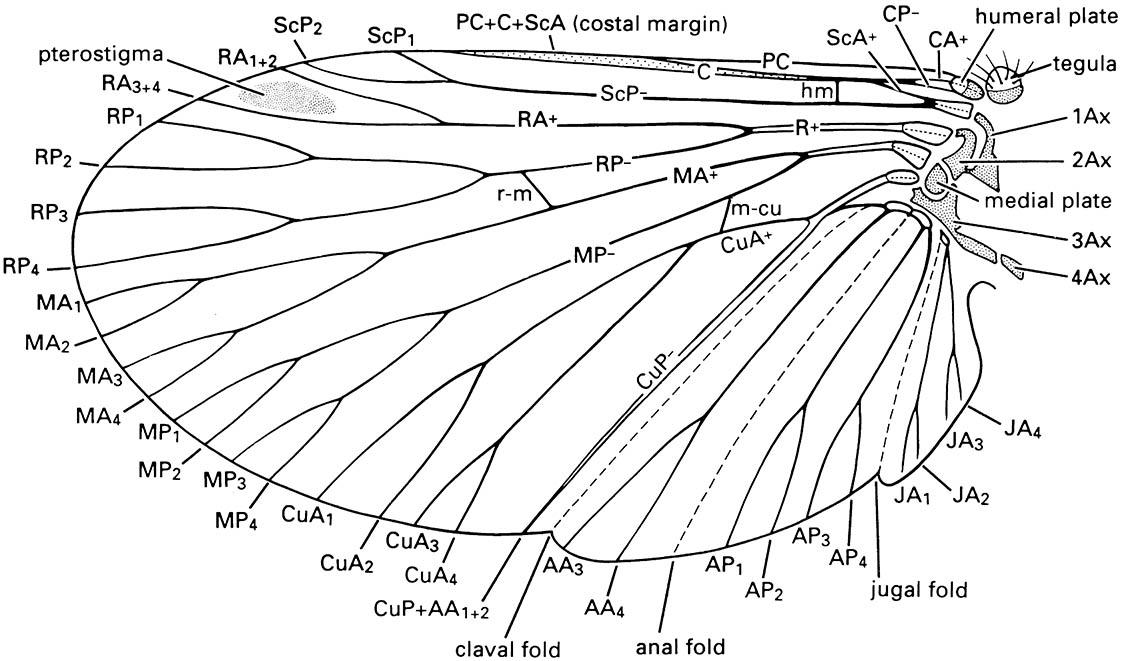
Notation as follows: AA, anal anterior; AP, anal posterior; Ax, axillary sclerite; C, costa; CA, costa anterior; CP, costa posterior; CuA, cubitus anterior; CuP, cubitus posterior; hm, humeral vein; JA, jugal anterior; MA, media anterior; m-cu, cross-vein between medial and cubital areas; MP, media posterior; PC, precosta; R, radius; RA, radius anterior; r-m, cross-vein between radial and median areas; RP, radius posterior; ScA, subcosta anterior; ScP, subcosta posterior. Branches of the anterior and posterior sector of each vein are numbered, e.g. CuA 1-4. (After CSIRO 1991)

