16.5.2. Microbial control
Microorganisms include bacteria, viruses, and small eukaryotes (e.g. protists, fungi, and nematodes). Some are pathogenic, usually killing insects, and of these many are host-specific to a particular insect genus or family. Infection is from spores, viral particles, or organisms that persist in the insect’s environment, often in the soil. These pathogens enter insects by several routes. Entry via the mouth (per os) is common for viruses, bacteria, nematodes, and protists. Cuticular and/or wound entry occurs in fungi and nematodes; the spiracles and anus are other sites of entry. Viruses and protists also can infect insects via the female ovipositor or during the egg stage. The microorganisms then multiply within the living insect but have to kill it to release more infectious spores, particles or, in the case of nematodes, juveniles. Disease is common in dense insect populations (pest or non-pest) and under environmental conditions suitable to the microorganisms. At low host density, however, disease incidence is often low as a result of lack of contact between the pathogens and their insect hosts.
Microorganisms that cause diseases in natural or cultured insect populations can be used as biological control agents in the same way as other natural enemies (section 16.5.1). The usual strategies of control are appropriate, namely:
- classical biological control (i.e. an introduction of an exotic pathogen such as the bacterium Paenibacillus (formerly Bacillus) popilliae established in the USA for the control of the Japanese beetle Popillia japonica (Scarabaeidae));
- augmentation via either:
- (i) inoculation (e.g. a single treatment that provides season-long control, as in the fungus Verticillium lecanii used against Myzus persicae aphids in glass- houses), or
- (ii) inundation (i.e. entomopathogens such as Bacillus thuringiensis used as microbial insecticides; see here and here);
- conservation of entomopathogens through manipulation of the environment (e.g. raising the humidity to enhance the germination and spore viability of fungi).
Some disease organisms are fairly host-specific (e.g. viruses) whereas others, such as fungal and nematode species, often have wide host ranges but possess different strains that vary in their host adaptation. Thus, when formulated as a stable microbial insecticide, different species or strains can be used to kill pest species with little or no harm to non-target insects. In addition to virulence for the target species, other advantages of microbial insecticides include their compatibility with other control methods and the safety of their use (non- toxic and non-polluting). For some entomopathogens (insect pathogens) further advantages include the rapid onset of feeding inhibition in the host insect, stability and thus long shelf-life, and often the ability to self-replicate and thus persist in target populations.
Obviously, not all of these advantages apply to every pathogen; many have a slow action on host insects, with efficacy dependent on suitable environmental conditions (e.g. high humidity or protection from sun- light) and appropriate host age and/or density. The very selectivity of microbial agents also can have practical drawbacks as when a single crop has two or more unrelated pest species, each requiring separate microbial control. All entomopathogens are more expensive to produce than chemicals and the cost is even higher if several agents must be used. However, bacteria, fungi, and nematodes that can be mass-produced in liquid fermenters (in vitro culture) are much cheaper to produce than those microorganisms (most viruses and protists) requiring living hosts (in vivo techniques). Some of the problems with the use of microbial agents are being overcome by research on formulations and mass- production methods.
Insects can become resistant to microbial pathogens as evidenced by the early success in selecting honey bees and silkworms resistant to viral, bacterial, and protist pathogens. Furthermore, many pest species exhibit significant intraspecific genetic variability in their responses to all major groups of pathogens. The current rarity of significant field resistance to microbial agents probably results from the limited exposure of insects to pathogens rather than any inability of most pest insects to evolve resistance. Of course, unlike chemicals, pathogens do have the capacity to coevolve with their hosts and over time there is likely to be a constant trade-off between host resistance, pathogen virulence, and other factors such as persistence.
Each of the five major groups of microorganisms (viruses, bacteria, protists, fungi, and nematodes) has different applications in insect pest control. Insecticides based on the bacterium Bacillus thuringiensis have been used most widely, but entomopathogenic fungi, nematodes, and viruses have specific and often highly successful applications. Although protists, especially microsporidia such as Nosema, are responsible for natural disease outbreaks in many insect populations and can be appropriate for classical biological control, they have less potential commercially than other micro-organisms because of their typical low pathogenicity (infections are chronic rather than acute) and the present difficulty of large-scale production for most species.
Nematodes
Nematodes from four families, the Mermithidae, Heterorhabditidae, Steinernematidae, and Neotylenchidae, include useful or potentially useful control agents for insects. The infective stages of entomopathogenic nematodes are usually applied inundatively, although establishment and continuing control is feasible under particular conditions. Genetic engineering of nematodes is expected to improve their biological control efficacy (e.g. increased virulence), production efficiency, and storage capacity. However, entomopathogenic nematodes are susceptible to desiccation, which restricts their use to moist environments.
Mermithid nematodes are large and infect their host singly, eventually killing it as they break through the cuticle. They kill a wide range of insects, but aquatic larvae of black flies and mosquitoes are prime targets for biological control by mermithids. A major obstacle to their use is the requirement for in vivo production, and their environmental sensitivity (e.g. to temperature, pollution, and salinity).
Heterorhabditids and steinernematids are small, soil-dwelling nematodes, associated with symbiotic gut bacteria (of the genera Photorhabdus and Xenorhabdus) that are pathogenic to host insects, killing them by septicemia. In conjunction with their respective bacteria, nematodes of Heterorhabditis and Steinernema can kill their hosts within two days of infection. They can be mass-produced easily and cheaply and applied with conventional equipment, and have the advantage of being able to search for their hosts. The infective stage is the third-stage juvenile (or dauer stage) — the only stage found outside the host. Host location is an active response to chemical and physical stimuli. Although these nematodes are best at controlling soil pests, some plant-boring beetle and moth pests can be controlled as well. Mole crickets (Gryllotalpidae: Scapteriscus spp.) are soil pests that can be infected with nematodes by being attracted to acoustic traps containing infective-phase Steinernema scapterisci, and then being released to inoculate the rest of the cricket population.
The Neotylenchidae contains the parasitic Deladenus siricidicola, which is one of the biological control agents of the sirex wood wasp, Sirex noctilio — a serious pest of forestry plantations of Pinus radiata in Australia. The juvenile nematodes infect larvae of S. noctilio, leading to sterilization of the resulting adult female wasp. This nematode has two completely different forms — one with a parasitic life cycle completely within the sirex wood wasp and the other with a number of cycles feeding within the pine tree on the fungus introduced by the ovipositing wasp. The fungal feeding cycle of D. siricidicola is used to mass culture the nematode and thus obtain infective juvenile nematodes for classical biological control purposes.
Fungi
Fungi are the commonest disease organisms in insects, with approximately 750 species known to infect arthropods, although only a few dozen naturally infect agriculturally and medically important insects. Fungal spores that contact and adhere to an insect germinate and send out hyphae. These penetrate the cuticle, invade the hemocoel and cause death either rapidly owing to release of toxins, or more slowly owing to massive hyphal proliferation that disrupts insect body functions. The fungus then sporulates, releasing spores that can establish infections in other insects; and thus the fungal disease may spread through the insect population.
Sporulation and subsequent spore germination and infection of entomopathogenic fungi often require moist conditions. Although formulation of fungi in oil improves their infectivity at low humidity, water requirements may restrict the use of some species to particular environments, such as soil, glasshouses, or tropical crops. Despite this limitation, the main advantage of fungi as control agents is their ability to infect insects by penetrating the cuticle at any developmental stage. This property means that insects of all ages and feeding habits, even sap-suckers, are susceptible to fungal disease. However, fungi can be difficult to mass-produce, and the storage life of some fungal products can be limited unless kept at low temperature. A novel application method uses felt bands containing living fungal cultures applied to the tree trunks or branches, as is done in Japan using a strain of Beauveria brongniartii against longhorn beetle borers in citrus and mulberry. Useful species of entomopathogenic fungi belong to genera such as Beauveria, Entomophthora, Hirsutella, Metarhizium, Nomuraea, and Verticillium. Many of these fungi overcome their hosts after very little growth in the insect hemocoel, in which case toxins are believed to cause death.
Verticillium lecanii is used commercially to control aphids and scale insects in European glasshouses. Entomophthora species also are useful for aphid control in glasshouses. Species of Beauveria and Metarhizium, known as white and green muscardines, respectively (depending on the color of the spores), are pathogens of soil pests, such as termites and beetle larvae, and can affect other insects, such as spittle bugs of sugarcane and certain moths that live in moist microhabitats. One Metarhizium species, M. anisopliae (= flavoviride) var. acridum, has been developed as a successful myco-insecticide for locusts and other grasshoppers in Africa.
Bacteria
Bacteria rarely cause disease in insects, although saprophytic bacteria, which mask the real cause of death, frequently invade dead insects. Relatively few bacteria are used for pest control, but several have proved to be useful entomopathogens against particular pests. Paenibacillus popilliae is an obligate pathogen of scarab beetles (Scarabaeidae) and causes milky disease (named for the white appearance of the body of infected larvae). Ingested spores germinate in the larval gut and lead to septicemia. Infected larvae and adults are slow to die, which means that P. popilliae is unsuitable as a microbial insecticide, but the disease can be transmitted to other beetles by spores that persist in the soil. Thus, P. popilliae is useful in biological control by introduction or inoculation, although it is expensive to produce. Two species of Serratia are responsible for amber disease in the scarab Costelytra zealandica, a pest of pastures in New Zealand, and have been developed for scarab control. Bacillus sphaericus has a toxin that kills mosquito larvae. The strains of Bacillus thuringiensis have a broad spectrum of activity against larvae of many species of Lepidoptera, Coleoptera, and aquatic Diptera, but can be used only as inundative insecticides because of lack of persistence in the field.
Bacillus thuringiensis, usually called Bt, was isolated first from diseased silkworms (Bombyx mori) by a Japanese bacteriologist, S. Ishiwata, about a century ago. He deduced that a toxin was involved in the pathogenicity of Bt and, shortly afterwards, other Japanese researchers demonstrated that the toxin was a protein present only in sporulated cultures, was absent from culture filtrates, and thus was not an exotoxin. Of the many isolates of Bt, several have been commercialized for insect control. Bt is produced in large liquid fermenters and formulated in various ways, including as dusts and granules that can be applied to plants as aqueous sprays. Currently, the largest market for Bt-based products (other than in transgenic plants) is the North American forestry industry.
Bt forms spores, each containing a proteinaceous inclusion called a crystal, which is the source of the toxins that cause most larval deaths. The mode of action of Bt varies among different susceptible insects. In some species insecticidal action is associated with the toxic effects of the crystal proteins alone (as for some moths and black flies). However, in many others (including a number of lepidopterans) the presence of the spore enhances toxicity substantially, and in a few insects death results from septicemia following spore germination in the insect midgut rather than from the toxins. For insects affected by the toxins, paralysis occurs in mouthparts, the gut, and often the body, so that feeding is inhibited. Upon ingestion by a larval insect, the crystal is dissolved in the midgut, releasing proteins called delta-endotoxins. These proteins are protoxins that must be activated by midgut proteases before they can interact with gut epithelium and disrupt its integrity, after which the insect ultimately dies. Early-instar larvae generally are more susceptible to Bt than older larvae or adult insects.
Effective control of insect pests by Bt depends on the following factors:
- the insect population being uniformly young to be susceptible;
- active feeding of insects so that they consume a lethal dose;
- evenness of spraying of Bt;
- persistence of Bt, especially lack of denaturation by ultraviolet light;
- suitability of the strain and formulation of Bt for the insect target.
Different Bt isolates vary greatly in their insecticidal activity against a given insect species, and a single Bt isolate usually displays very different activity in different insects. At present there are about 80 recognized Bt subspecies (or serovars) based on serotype and certain biochemical and host-range data. There is dis- agreement, however, concerning the basis of the Bt classification scheme, as it may be more appropriate to use a system based on the crystal toxin genes, which directly determine the level and range of Bt activity. The nomenclature and classification scheme for crystal genes (cry) is based on their phenotype, types of crystal proteins produced, and the protein’s host range as insecticidal toxins. Toxins are encoded by the cryI, cryII, cryIII, cryIV and cyt, and cryV gene classes: cryI genes are associated with bipyramidal crystals that are toxic to lepidopteran larvae; cryII with cuboidal crystals active against both lepidopteran and dipteran larvae; cryIII with flat, square crystals toxic to coleop- teran larvae; cryIV and cyt with various-shaped crys- tals that kill dipteran larvae; and cryV, which is toxic to lepidopteran and some coleopteran larvae. B. t. israelensis, for example, has cryIV and cyt genes, whereas B. t. tenebrionis has cryIII genes, and B. t. kurstaki has cryI and cryII genes. In addition, some cultures of Bt produce exotoxins, which are effective against various insects including larvae of the Colorado potato beetle.
Thus, the nature and insecticidal effects of the various isolates of Bt are far from simple and further research on the modes of action of the toxins is desirable, especially for understanding the basis of potential and actual resistance to Bt.
Bt products have been used increasingly for control of various Lepidoptera (such as caterpillars on crucifers and in forests) since 1970. For the first two decades of use, resistance was rare or unknown, except in a stored-grain moth (Pyralidae: Plodia interpunctella). The first insect to show resistance in the field was a major plant pest, the diamondback moth (Plutellidae: Plutella xylostella), which is believed to be native to South Africa. Watercress growers in Japan and Hawai’i complained that Bt had reduced ability to kill this pest, and by 1989 further reports of resistant moths in Hawai’i were confirmed in areas where frequent high doses of Bt had been used. Similarly in Japan, by 1988 an extremely high level of Bt resistance was found in moths in greenhouses where watercress had been grown year-round with a total of 40–50 applications of Bt over three to four years. Moths resistant to Bt also were reported in Thailand, the Philippines, and main- land USA. Furthermore, laboratory studies and field reports have indicated that more than a dozen other insect species have naturally evolved or could be bred to show differing levels of resistance. Bt resistance mechanisms of the diamondback moth have been shown to derive from a single gene that confers resistance to four different Bt toxins.
Problems with chemical insecticides have stimulated interest in the use of Bt products as an alternative method of pest control. In addition to conventional applications of Bt, genetic engineering with Bt genes has produced transgenic plants (“Bt plants”) that manufacture their own protective toxins (section 16.6.1), such as INGARD cotton, which carries the cryIA(c) Bt gene, and transgenic varieties of corn and soybean that are grown widely in the USA. Current optimism has led to the belief that insects are unlikely to develop extremely high levels of Bt resistance in the field, as a result of both instability of resistance and dilution by immigrants from susceptible populations. Strategies to prevent or slow down the evolution of resistance to Bt are the same as those used to retard resistance to synthetic insecticides. Obviously, the continued success of Bt products and the benefits of technological advances will depend on appropriate use as well as understanding and limiting resistance to the Bt crystal proteins.
Viruses
Many viruses infect and kill insects, but those with potential for insect control are from just three viral groups, all with proteinaceous inclusion bodies, which enclose the virions (virus particles). These “occluded” viral species are considered safe because they have been found only in arthropods and appear unable to replicate in vertebrates or vertebrate cell cultures, although distant relatives of two of these groups have wider host ranges. Many “non-occluded” viruses that infect insects are considered unsafe for pest control because of their lack of specificity and possible adverse side-effects (such as infection of vertebrates and/or beneficial insects).
The useful entomopathogenic groups are the nuclear polyhedrosis viruses (NPVs), granulosis viruses (GVs) (both belonging to Baculoviridae — the baculoviruses or BVs), the cytoplasmic polyhedrosis viruses (CPVs) (Reoviridae: Cypovirus), and the ento-mopoxviruses (EPVs) (Poxviridae: Entomopoxvirinae). Baculoviruses replicate within the nuclei of the host cells, whereas the CPVs and EPVs replicate in the host cell cytoplasm. Baculoviruses have DNA genomes and are found mostly in endopterygotes, such as moth and beetle larvae, which become infected when they ingest the inclusion bodies with their food. Inclusion bodies dissolve in the high pH of the insect midgut and release the virion(s) (Fig. 16.4). These infect the gut epithelial cells and usually spread to other tissues, particularly the fat body. The inclusion bodies of NPVs are usually very stable and may persist in the environment for years (if protected from ultraviolet light, as in the soil), increasing their utility as biological control agents or microbial insecticides. The host-specificity of different viruses also influences their potential usefulness as pest control agents; some baculoviruses (such as the Helicoverpa NPV) are specific to an insect genus. CPVs have RNA genomes and have been found in more than 200 insect species, mainly of Lepidoptera and Diptera. Their inclusion bodies are less stable than those of NPVs. EPVs have large DNA genomes and infect a wide range of hosts in the Orthoptera, Lepidoptera, Coleoptera, and Diptera, but individual viral isolates generally have a narrow host range. Infection of insect cells follows a similar path to that of baculoviruses.
For certain pests, viral insecticides provide feasible alternatives to chemical controls but several factors may restrict the usefulness of different viruses. Ideally, viral insecticides should be host-specific, virulent, kill quickly, persist for a reasonable time in the environment after application, and be easy to provide in large amounts. CPVs fulfill these requirements poorly, whereas the other viruses score better on these criteria, although they are inactivated by ultraviolet light within hours or days, often they kill larvae slowly and/or have a low virulence, and production costs can be high. At present, viral pesticides are produced mostly by in vivo or small-scale in vitro methods, which are expensive because of the costs of rearing the host larvae; although an in vivo technology called HeRD (high efficiency rearing device) greatly improves the cost/benefit ratio for producing baculovirus pesticide. Also, the use of new tissue culture technology has significantly reduced the very high cost of in vitro production methods. Potency problems may be overcome by genetic engineering to increase either the speed of action or the virulence of naturally occurring viruses, such as the baculoviruses that infect the heliothine pests (Lepidoptera: Noctuidae: Helicoverpa and Heliothis spp.) of cotton. The presence of particular proteins appears to enhance the action of baculoviruses; viruses can be altered to produce much more protein or the gene controlling protein production can be added to viruses that lack it. There is considerable commercial interest in the manufacture of toxin-producing viral insecticides by inserting genes encoding insecticidal products, such as insect-specific neurotoxins, into baculoviruses. However, the environmental safety of such genetically engineered viruses must be evaluated carefully prior to their wide-scale application.
Insect pests that damage valuable crops, such as boll-worms of cotton and sawflies of coniferous forest trees, are suitable for viral control because substantial economic returns offset the large costs of development (including genetic engineering) and production. The other way in which insect viruses could be manipulated for use against pests is to transform the host plants so that they produce the viral proteins that damage the gut lining of phytophagous insects. This is analogous to the engineering of host-plant resistance by incorporating foreign genes into plant genomes using the crown-gall bacterium as a vector (section 16.6.1).
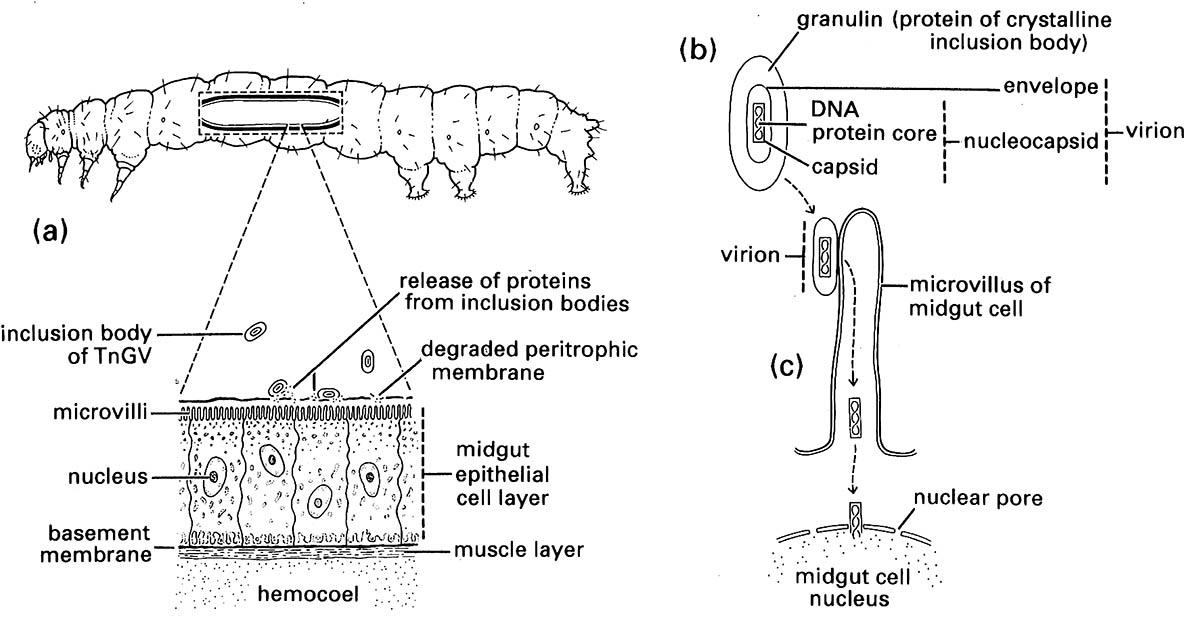
(a) A caterpillar of the cabbage looper, Trichoplusiani (Lepidoptera: Noctuidae), ingests the viral inclusion bodies of a granulosis virus (called TnGV) with its food and the inclusion bodies dissolve in the alkaline midgut releasing proteins that destroy the insect’s peritrophic membrane, allowing the virions access to the midgut epithelial cells. (b) A granulosis virus inclusion body with virion in longitudinal section. (c) A virion attaches to a microvillus of a midgut cell, where the nucleocapsid discards its envelope, enters the cell and moves to the nucleus in which the viral DNA replicates. The newly synthesized virions then invade the hemocoel of the caterpillar where viral inclusion bodies are formed in other tissues (not shown). (After Entwistle & Evans 1985; Beard 1989)

