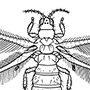
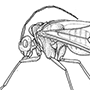
Box 11.8. Hemiptera (bugs, cicadas, leafhoppers, spittle bugs, planthoppers, aphids, jumping plant lice, scale insects, whiteflies)
The Hemiptera is distributed worldwide, and is the most diverse of the non-endopterygote orders, with more than 90,000 species in about 140 families. Historically, it was divided into two suborders (sometimes treated as orders): Heteroptera (bugs) and “Homoptera” (cicadas, leafhoppers, spittle bugs, planthoppers, aphids, jump- ing plant lice (= psylloids), scale insects (= coccoids), and whiteflies). However, homopterans represent a grade of organization (a paraphyletic rather than a monophyletic group). Currently, five suborders can be recognized (Fig. 7.5):
- (i) Heteroptera, the “true” bugs;
- (ii) Coleorrhyncha, the moss bugs (family Peloridiidae);
- (iii) Cicadomorpha (cicadas, leafhoppers, and spittle bugs);
- (iv) Fulgoromorpha (planthoppers); and
- (v) Sternorrhyncha (aphids, jumping plant lice, scale insects, and whiteflies).
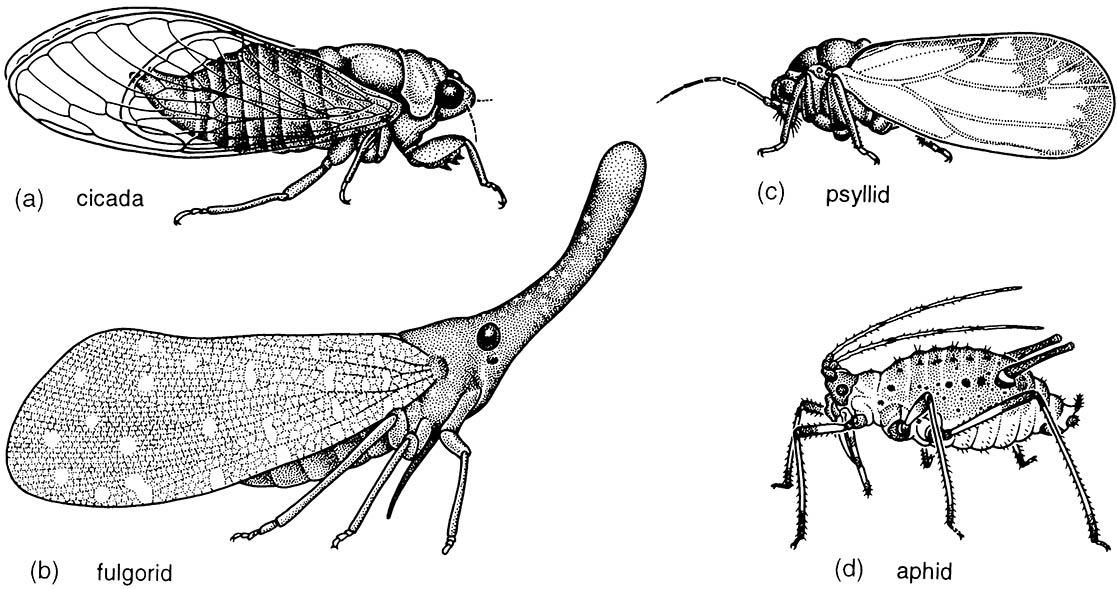
Sometimes the Cicadomorpha and Fulgoromorpha are collectively called the Auchenorrhyncha, but this grouping may also be paraphyletic. Four hemipterans are illustrated here in lateral view: (a) Cicadetta montana (Cicadidae), the only British cicada (after drawing by Jon Martin in Dolling 1991); (b) a green lantern bug, Pyrops sultan (Fulgoridae), from Borneo (after Edwards 1994); (c) the psyllid Psyllopsis fraxini (Psyllidae), which deforms leaflets of ash trees in Britain (after drawing by Jon Martin in Dolling 1991); and (d) an apterous viviparous female of the aphid Macromyzus woodwardiae (Aphididae) (after Miyazaki 1987a).
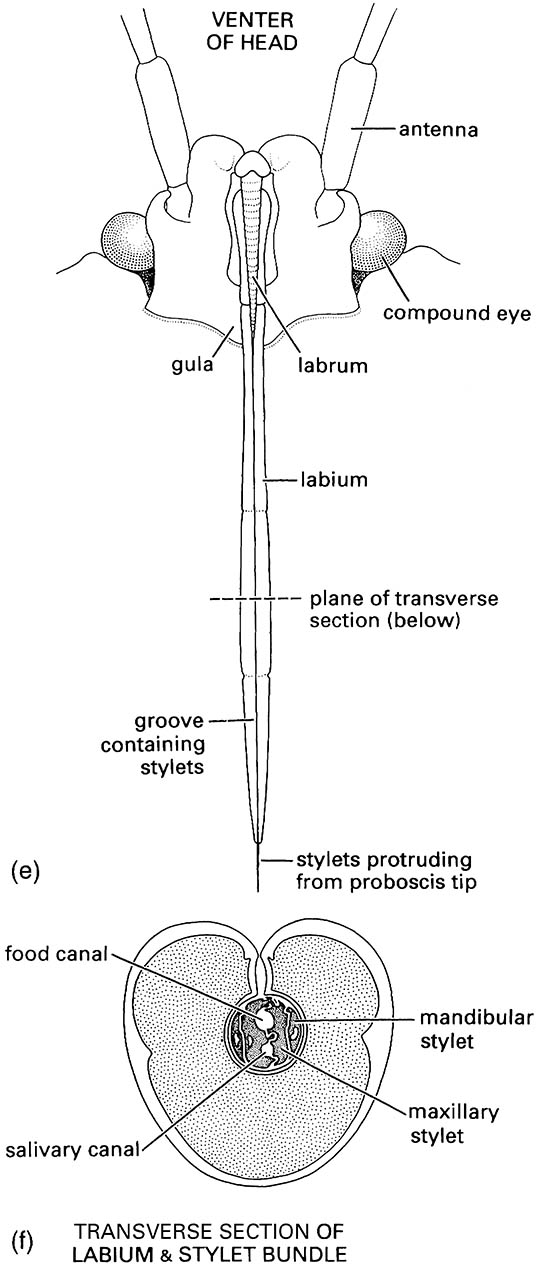
Hemipteran compound eyes are often large, and ocelli may be present or absent. Antennae vary from short with few segments to filiform and multiseg- mented. The mouthparts comprise mandibles and maxillae modified as needle-like stylets, lying in a beak-like grooved labium (as shown for a pentatomid heteropteran in (e) and (f )), collectively forming a rostrum or proboscis. The stylet bundle contains two canals, one delivering saliva, the other uptaking fluid (as shown in (f )); there are no palps. The thorax often consists of large pro- and mesothorax, but a small metathorax. Both pairs of wings often have reduced venation; some hemipterans are apterous, and rarely there may be just one pair of wings (in male scale insects). The legs are frequently gressorial, sometimes raptorial, often with complex pretarsal adhesive structures. The abdomen is variable, and cerci are absent.
Most Heteroptera hold their head horizontally, with the rostrum anteriorly distinct from the prosternum (although the rostrum may be in body contact at coxal bases and on anterior abdomen). When at rest the wings are usually folded flat over the abdomen (Fig. 5.8). The fore wings usually are thickened basally and membranous apically to form hemelytra (Fig. 2.22e). Heteroptera mostly have abdominal scent glands. Apterous heteropterans can be identified by the rostrum arising from the anteroventral region of the head and the presence of a large gula. Non-heteropterans hold the head deflexed with the complete length of the rostrum appressed to the prosternum, directed posteriorly often between the coxal bases. They have membranous wings that rest roof-like over the abdomen; apterous species are identified by the absence of a gula and the rostrum arising from the posteroventral head or near the prosternum. Mouthparts are absent in some aphids, and in some female and all male scale insects.
Nymphal Heteroptera (Fig. 6.2) resemble adults except in the lack of development of the wings and genitalia. However, immature Sternorrhyncha show much variation in a range of complex life cycles. Many aphids exhibit parthenogenesis (section 5.10.1), usually alternating with seasonal sexual reproduction. The immature stages of Aleyrodoidea (whiteflies) and Coccoidea (scale insects) may differ greatly from adults, with larviform stages followed by a quiescent, non-feeding “pupal” stage, in convergently acquired holometaboly.
The primitive feeding mode is piercing and sucking plant tissue (Fig. 11.4), and many species induce galls on their host plants (section 11.2.4; Box 11.2). All hemipterans have large salivary glands and an alimentary canal modified for absorption of liquids, with a filter chamber to remove water (Box 3.3). Many hemipterans rely exclusively on living plant sap (from phloem or xylem and sometimes parenchyma). Elimination of large quantities of honeydew by phloem-feeding Sternorrhyncha provides the basis for mutualistic relationships with ants. Many hemipterans exude waxes (Fig. 2.5), which form powdery (see Plate 5.1, facing here) or plate-like protective covers. Non-phytophagous Heteroptera comprise many predators, some scav- engers, a few hematophages (blood-feeders), and some necrophages (consumers of dead prey), with the last trophic group including successful colonizers of aquatic environments (Box 10.6) and some of the few insects to live on the oceans (section 10.8).
Phylogenetic relationships are considered in section 7.4.2 and depicted in Fig. 7.5.
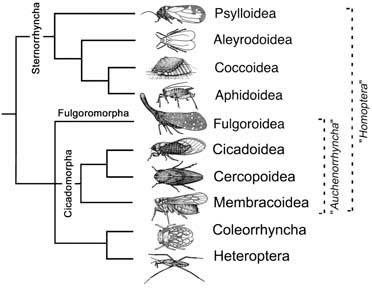
Broken lines indicate paraphyletic taxa, with names italicized. (After Bourgoin & Campbell 2002)
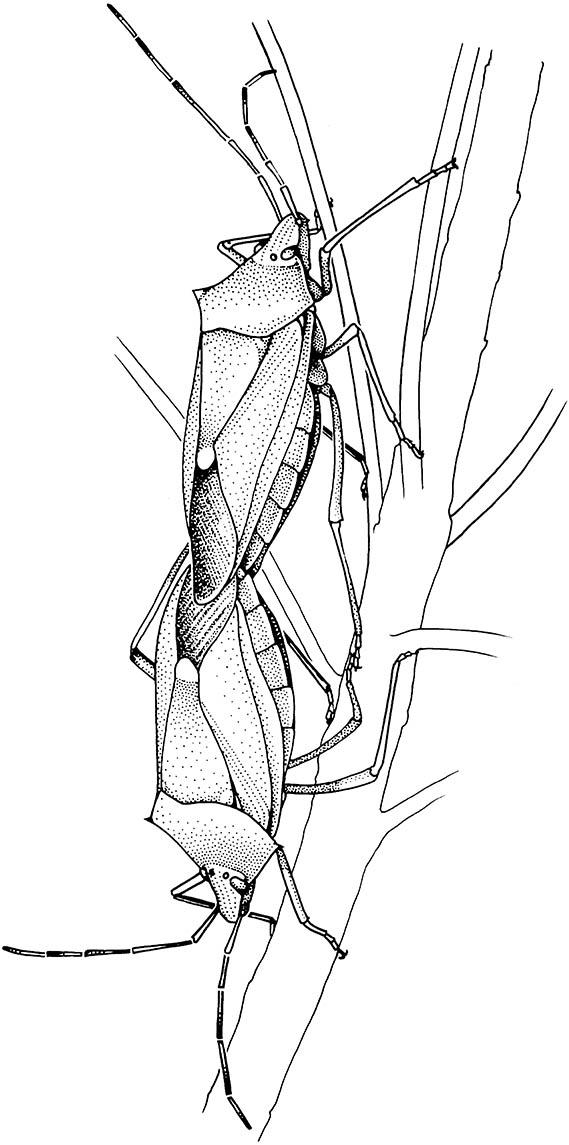
Many heteropteran bugs engage in prolonged copulation, which prevents other males from inseminating the female until either she becomes non-receptive to further males or she lays the eggs fertilized by the “guarding” male.
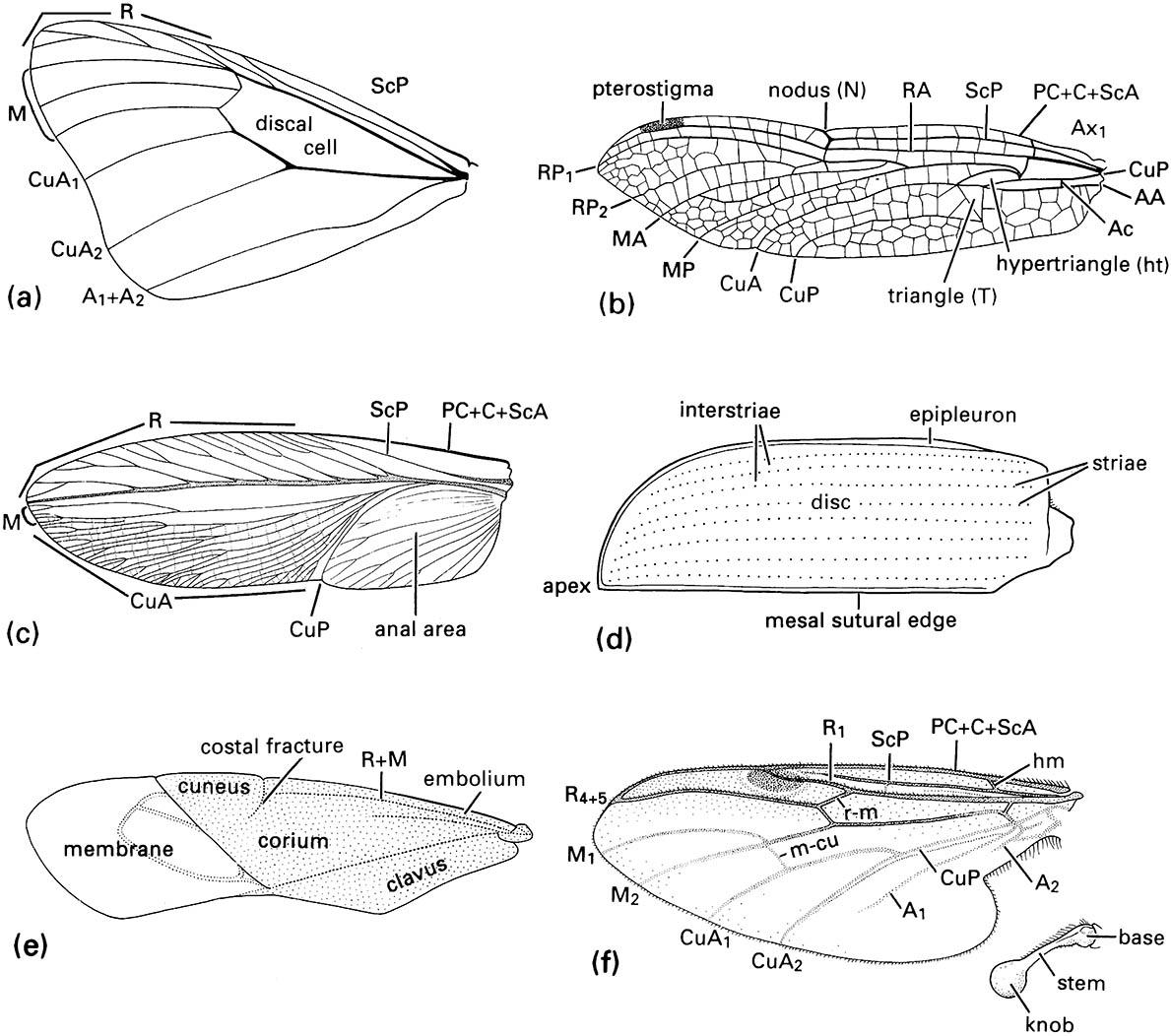
(a) fore wing of a butterfly of Danaus (Lepidoptera: Nymphalidae); (b) fore wing of a dragonfly of Urothemis (Odonata: Anisoptera: Libellulidae); (c) fore wing or tegmen of a cockroach of Periplaneta (Blattodea: Blattidae); (d) fore wing or elytron of a beetle of Anomala (Coleoptera: Scarabaeidae); (e) fore wing or hemelytron of a mirid bug (Hemiptera: Heteroptera: Miridae) showing three wing areas — the membrane, corium, and clavus; (f ) fore wing and haltere of a fly of Bibio (Diptera: Bibionidae) (after J.W.H. Trueman, unpublished. ((a-d) After Youdeowei 1977; (f) after McAlpine 1981)
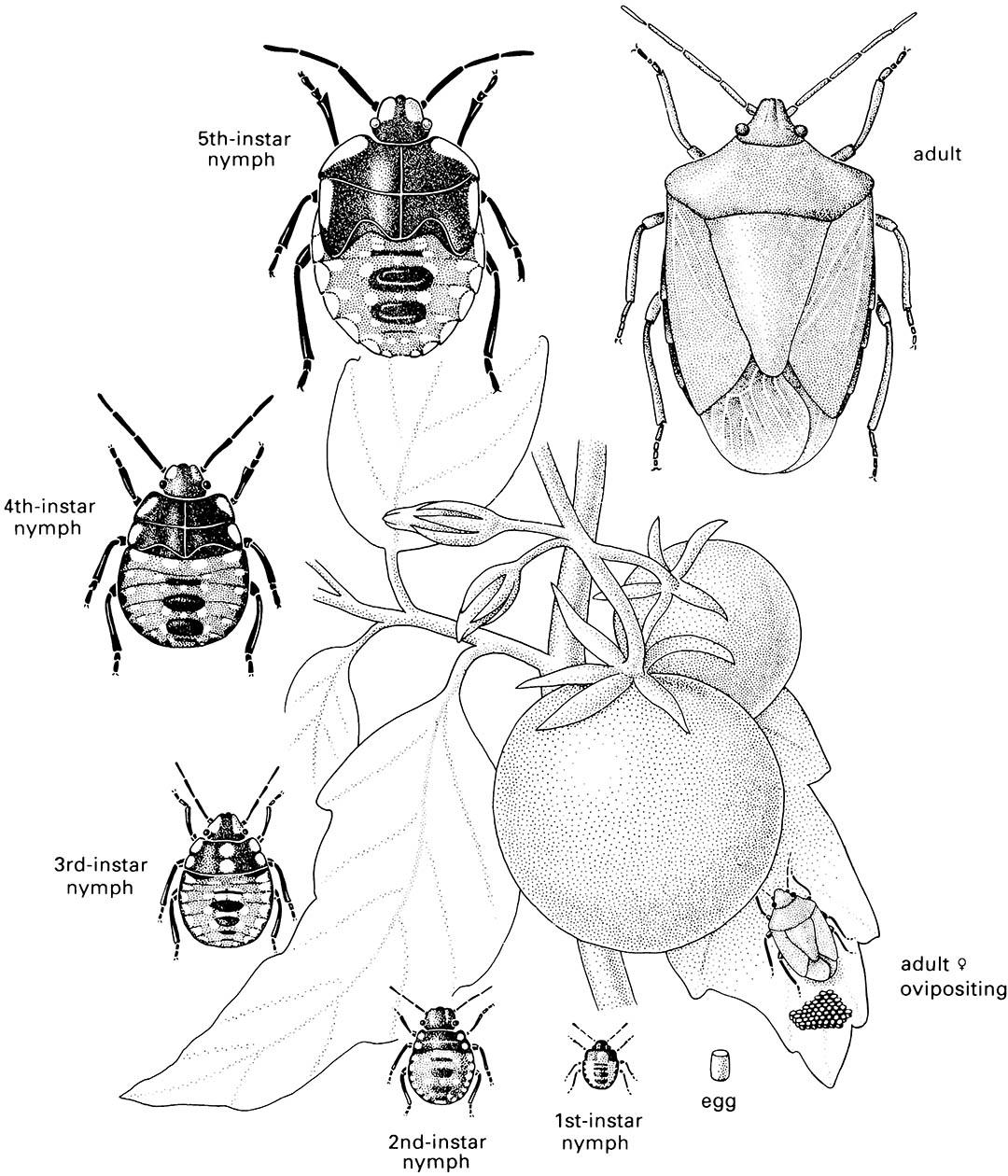
This cosmopolitan and polyphagous bug is an important world pest of food and fiber crops. (After Hely et al. 1982)
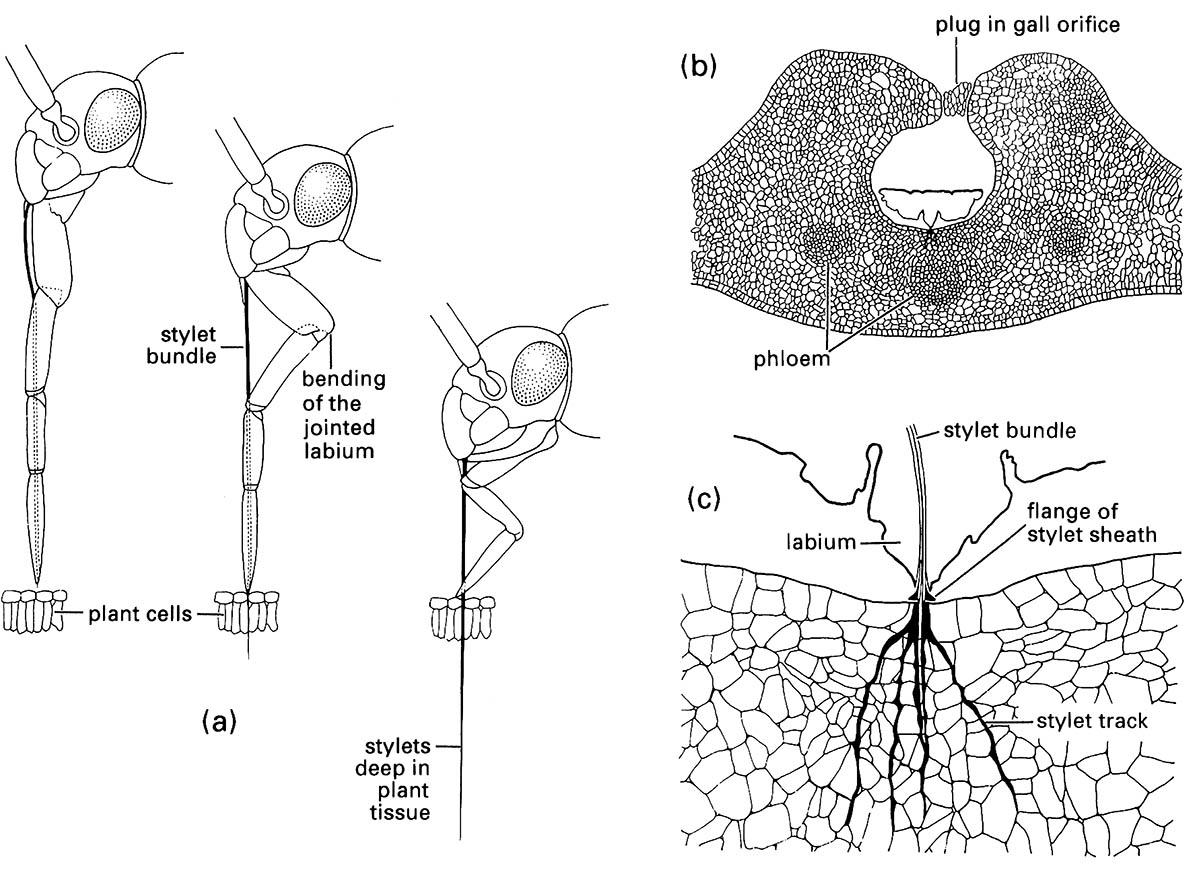
(a) penetration of plant tissue by a mirid bug showing bending of the labium as the stylets enter the plant; (b) transverse section through a eucalypt leaf gall containing a feeding nymph of a scale insect, Apiomorpha (Eriococcidae); (c) enlargement of the feeding site of (b) showing multiple stylet tracks (formed of solidifying saliva) resulting from probing of the parenchyma. ((a) After Poisson 1951)
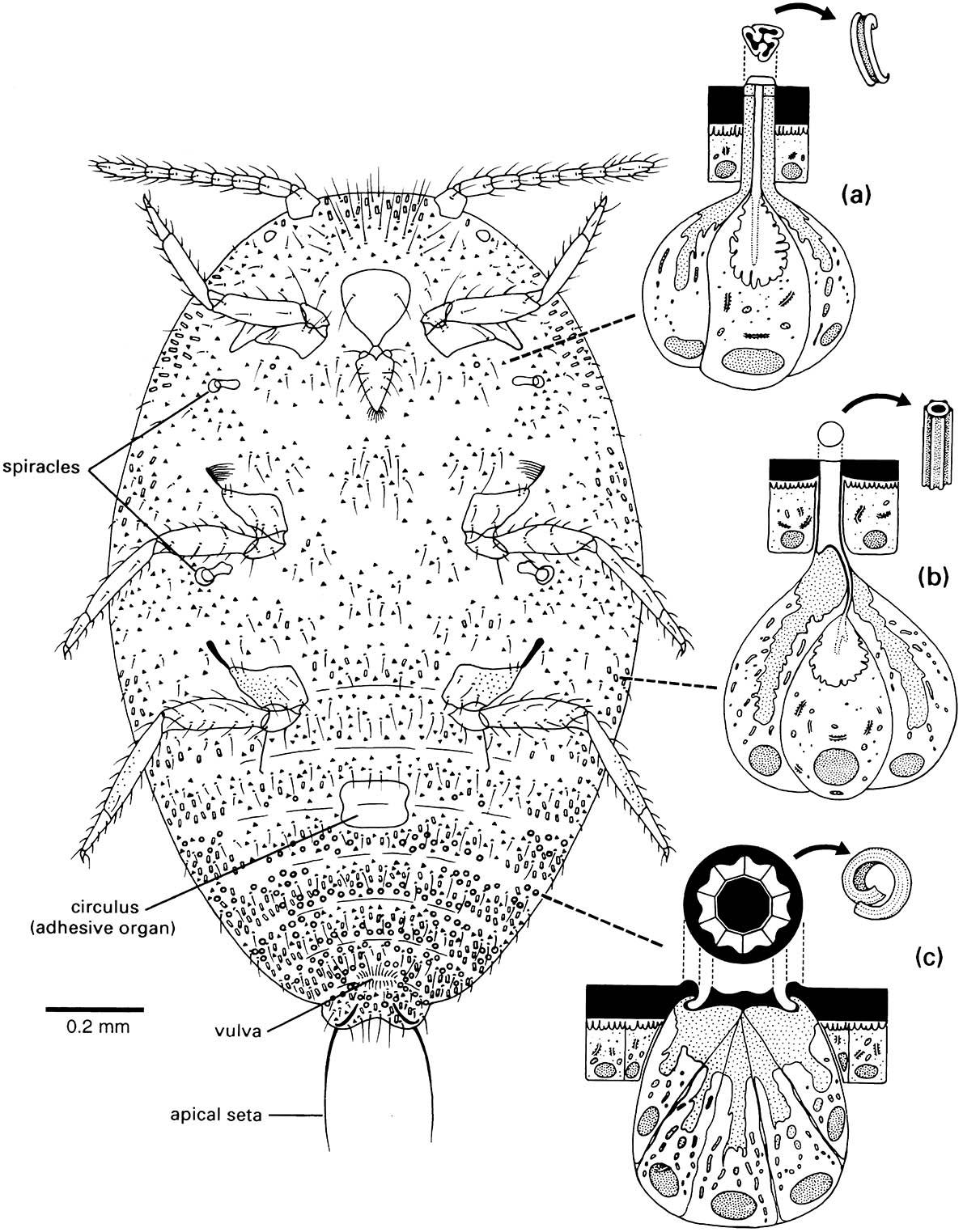
Enlargements depict the ultrastructure of the wax glands and the various wax secretions (arrowed) associated with three types of cuticular structure: (a) a trilocular pore; (b) a tubular duct; and (c) a multilocular pore. Curled filaments of wax from the trilocular pores form a protective body-covering and prevent contamination with their own sugary excreta, or honeydew; long, hollow, and shorter curled filaments from the tubular ducts and multilocular pores, respectively, form the ovisac. (After Foldi 1983; Cox 1987)