Box 11.2. The grape phylloxera
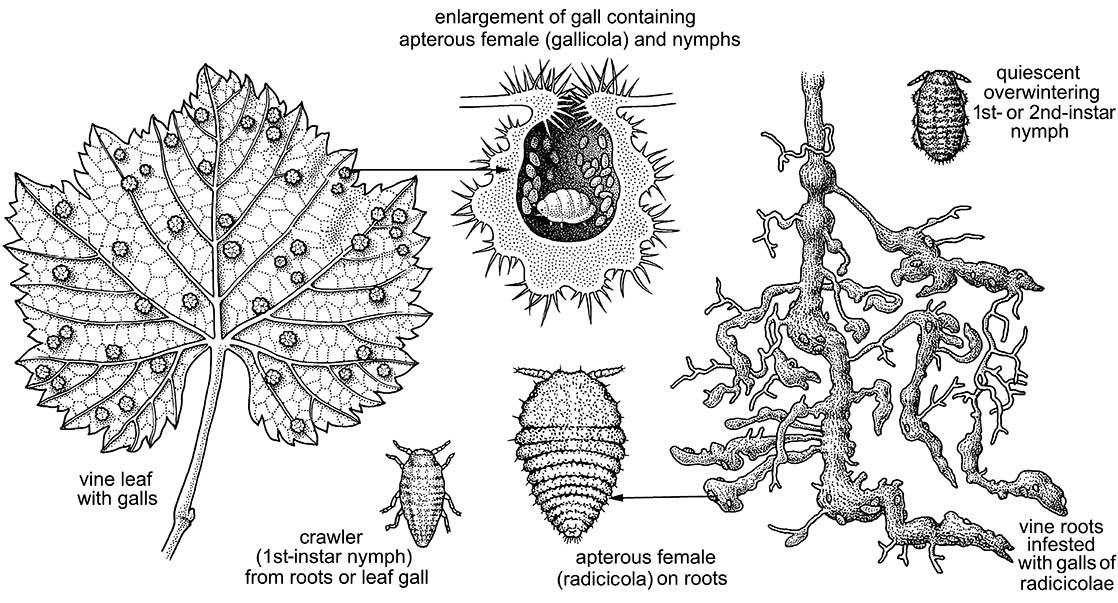
An example of the complexity of a galling life cycle, host-plant resistance, and even naming of an insect is provided by the grape phylloxera, sometimes called the grape louse. This aphid’s native range and host is temperate—subtropical from eastern North America and the south-west including Mexico, on a range of species of wild grapes (Vitaceae: Vitis spp.). Its complete life cycle is holocyclic (restricted to a single host). In its native range, its life cycle commences with the hatching of an overwintering egg, which develops into a fundatrix that crawls from the vine bark to a developing leaf where a pouch gall is formed in the rapidly growing meristematic tissue (as shown here, after several sources). Numerous generations of further apterous offspring are produced, most of which are gallicolae — gall inhabitants that either continue to use the maternal gall or induce their own. Some of the apterae, termed radicicolae, migrate downwards to the roots. In warm climate regions such as California, South Africa, and Australia where the phylloxera is introduced, it is radicicolae that survive the winter when vine leaves are shed along with their gallicolae. In the soil, radicicolae form nodose and tuberose galls (swellings) on the subapices of young roots (as illustrated here for the asexual life cycle). In fall, in those biotypes with sexual stages, alates (sexuparae) are produced that fly from the soil to the stems of the vine, where they give rise to apterous, non-feeding sexuales. These mate, and each female lays a single overwintering egg. Within the natural range of aphid and host, the plants appear to show little damage from phylloxera, except perhaps in the late season in which limited growth provides only a little new meristematic tissue for the explosive increase in gallicolae.
This straightforward (for an aphid) life cycle shows modifications outside the natural range, involving loss of the sexual and aerial stages, with persistence owing to entirely parthenogenetic radicicolae. Also involved are dramatic deleterious effects on the host vine by phylloxera feeding. This is of major economic importance when the host is Vitis vinifera, the native grape vine of the Mediterranean and Middle East. In the mid- 19th century American vines carrying phylloxera were imported into Europe; these devastated European grapes, which had no resistance to the aphid. Damage is principally through roots rotting under heavy loads of radicicolae rather than sucking perse, and generally there is no aerial gall-inducing stage. The shipment from eastern USA to France by Charles Valentine Riley of a natural enemy, the mite Tyroglyphus phylloxerae, in 1873 was the first intercontinental attempt to control a pest insect. However, eventual control was achieved by grafting the already very diverse range of European grape cultivars (cépages such as Cabernet, Pinot Noir, or Merlot) onto phylloxera-resistant rootstocks of North American Vitis species. Some Vitis species are not attacked by phylloxera, and in others the infestation starts and is either tolerated at a low level or rejected. Resistance (section 16.6) is mainly a matter of the speed at which the plant can produce inhibitory complex compounds from naturally produced phenolics that can isolate each developing tuberose gall. Recently it seems that some genotypes of phylloxera have circumvented certain resistant rootstocks, and resurgence may be expected.
The history of the scientific name of grape phylloxera is nearly as complicated as the life cycle — phylloxera may now refer only to the family Phylloxeridae, in which species of Phylloxera are mainly on Juglans (walnuts), Carya (pecans), and relatives. The grape phylloxera has been known as Phylloxera vitifoliae and also as Viteus vitifoliae (under which name it is still known in Europe), but it is increasingly accepted that the genus name should be Daktulosphaira if a separate genus is warranted. Whether there is a single species (D. vitifoliae) with a very wide range of behaviors associated with different host species and cultivars is an open question. There certainly is wide geographical variation in responses and host tolerances but as yet no morphometric, molecular, or behavioral traits correlate well with any of the reported “biotypes” of D. vitifoliae.